 The compounds with the larger lattice energy are Since the lattice energy is negative in the Born-Haber cycle, this would lead to a more exothermic reaction.Ĥ. 4008 kJ/mol both ions in MgO have twice the charge of the ions in LiF the bond length is very similar and both have the same structure a quadrupling of the energy is expected based on the equation for lattice energyĥ. The smaller the radius of the anion, the shorter the interionic distance and the greater the lattice energy would be. 
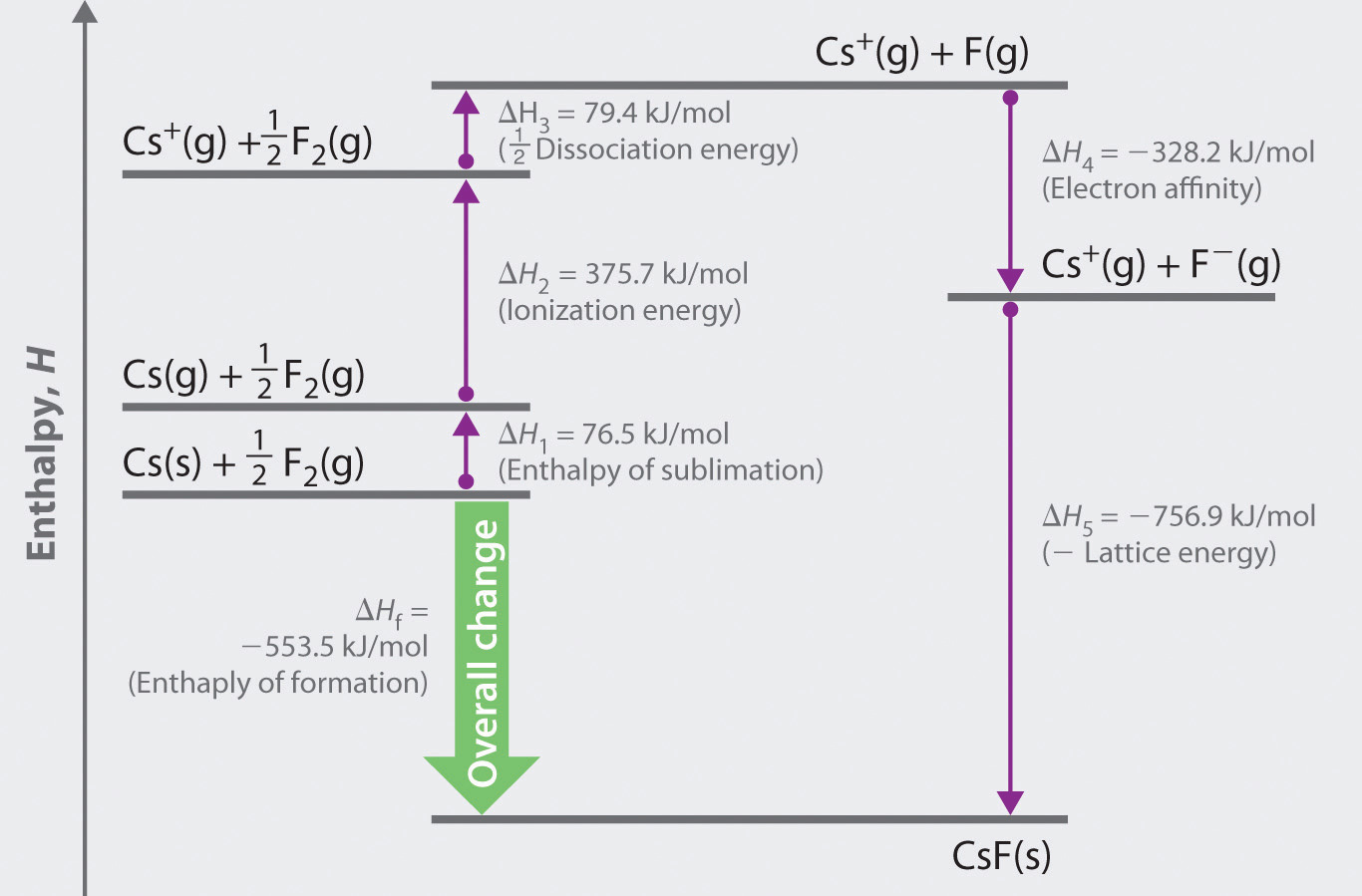
In the Born-Haber cycle, the more negative the electron affinity, the more exothermic the overall reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |




 RSS Feed
RSS Feed
