 
For example: rico Marlon de Moraes Flores. Once again, physics as an enabling science changed the way we live. Bibliographic details should be cited in the order: year, volume, page, and must include the article title. 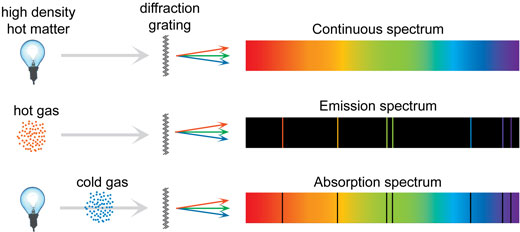
When this “ultraviolet catastrophe” was eventually solved, the answers led to new technologies such as computers and the sophisticated imaging techniques described in earlier chapters. Various orders of the spectrum are shown. In fact, providing a theoretical explanation for the experimentally measured shape of the spectrum was a mystery at the turn of the century. Figure 1: Schematic illustration of diffraction of light by a diffraction grating. To specify a state, one must list four quantum numbers for each electron, for example. All of this seems quite continuous, but it was the curve of the spectrum of intensity versus wavelength that gave a clue that the energies of the atoms in the solid are quantized. The state of an atom is the condition of motion of all the electrons. 
This figure shows an example of an emission spectrum obtained by passing an electric discharge through. It was discussed that the total intensity of the radiation varies as \(T^4\), the fourth power of the absolute temperature of the body, and that the peak of the spectrum shifts to shorter wavelengths at higher temperatures. Atomic spectra remain an important analytical tool today. The color a substance emits when its electrons get excited can be used to help identify which elements are present in a given sample. The following shows a few examples of the emission spectra for some common elements: The atomic emission spectra for H, He, N, O, Ar, Ne, Xe, and Hg. Ideal radiators are therefore called blackbodies, and their EM radiation is called blackbody radiation. These atomic spectra are almost like elements fingerprints. In forensic science, these techniques are of utmost importance. For example, an atom in its lowest possible energy state (called the. Atomic spectroscopy mainly consists of two types of techniques: atomic absorption spectroscopy and atomic emission spectroscopy. Atomic energy levels are typically measured by observing transitions between two levels. \)) An ideal radiator is one that has an emissivity of 1 at all wavelengths and, thus, is jet black. Atomic spectroscopy is an excellent analytical tool used for the detection and measurement of elements in a sample with high precision and confidence. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
